
Book a demo
A game-changing product that will simplify and enhance your workflow. Designed for companies looking to streamline their trial management processes.
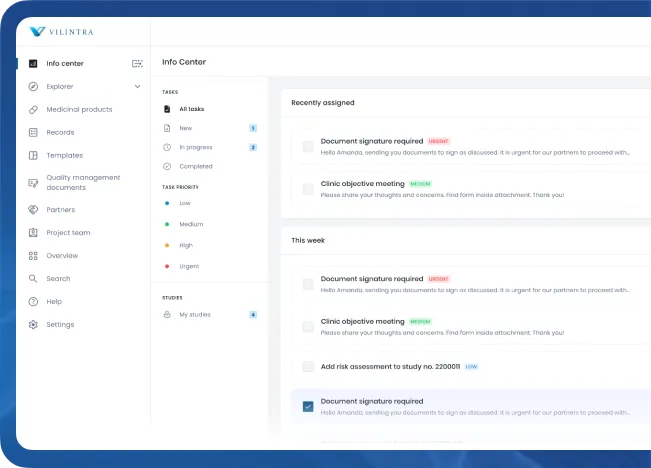
About us
Our mission is to simplify and enhance the complex workflows involved in trial management, ensuring that every step, from document generation to final archiving, is executed with precision and speed.
Developed with insights from industry leaders and tested in real-world settings since 2017, Vilintra stands out for its ability to significantly reduce human error, improve productivity, and ensure compliance with global standards like GAMP 5, GDPR, and FDA regulations. Our commitment to innovation and quality ensures that your clinical trials are conducted with the utmost reliability and efficiency, paving the way for faster time-to-market for life-saving medicines.
Frequently Asked Questions
Our solution is designed for pharmaceutical companies, clinical research organizations (CROs), and research institutions that manage complex clinical trials and need to streamline their trial management processes while ensuring compliance with regulatory standards.
Our solution significantly reduces the risk of human error by automatically generating documents, requiring only raw data input from users.
Our solution is hosted by a client so all documentation is stored locally and only you can access it. We are currently also developing a cloud-based solution.
Yes, Vilintra’s modular design allows you to choose and implement only the specific functions or modules that meet your current needs, whether it’s trial management, document generation, or monitoring.
Absolutely. Vilintra is highly customizable and can be tailored to fit the specific workflows, regulatory requirements, and operational needs of your organization, ensuring a solution that works for you.



